https://www.epfl.ch/labs/lcso/ https://www.linkedin.com/in/lcso-lab/
Starter packs - OrgChem https://go.bsky.app/NSEFPFJ https://go.bsky.app/GwH8t
- Chem. in CH https://go.bsky.app/CdvVFKj
go.bsky.app/GwH8tnF
(completing the already full OrgChem:
go.bsky.app/NSEFPFJ)

A formal (3+2)-cycloaddition of donor-acceptor cyclopropanes and ammonium thiocyanate

A formal (3+2)-cycloaddition of donor-acceptor cyclopropanes and ammonium thiocyanate
onlinelibrary.wiley....
#ChemSky #ChemBio

onlinelibrary.wiley....
#ChemSky #ChemBio
More fun with oxabicycles.
pubs.acs.org/doi/full/10....

More fun with oxabicycles.
pubs.acs.org/doi/full/10....
pubs.rsc.org/en/Content/A...
pubs.rsc.org/en/Content/A...
onlinelibrary.wiley.com/journal/1522...

onlinelibrary.wiley.com/journal/1522...
Article by Xile Hu & co-workers @xilehu-epfl.bsky.social
Construction of dual-cofactor artificial metalloenzymes for synergistic and enantiodivergent catalysis of Michael addition reactions
www.nature.com/articles/s44... ($)
#Chemsky

Check out the newest study by the group of @sebastianpomplun.bsky.social of our @led3hub.bsky.social. Being able to predict peptide binders solely based on structure may be closer than you think.
pubs.acs.org/doi/10.1021/...
#ChemSky #ChemBio

Check out the newest study by the group of @sebastianpomplun.bsky.social of our @led3hub.bsky.social. Being able to predict peptide binders solely based on structure may be closer than you think.
pubs.acs.org/doi/10.1021/...
#ChemSky #ChemBio
Excited to share our latest work on generative modelling for accelerating the discovery of organic photocatalysts💡✨
👉Read more: doi.org/10.26434/che...
Congratulations to Leon Schlosser, Nils Rendel, Julius Gemen, @gloriusfrank.bsky.social, and @valencekjell.com

Excited to share our latest work on generative modelling for accelerating the discovery of organic photocatalysts💡✨
👉Read more: doi.org/10.26434/che...
Congratulations to Leon Schlosser, Nils Rendel, Julius Gemen, @gloriusfrank.bsky.social, and @valencekjell.com


onlinelibrary.wiley.com/doi/10.1002/...

onlinelibrary.wiley.com/doi/10.1002/...
pubs.acs.org/doi/10.1021/...
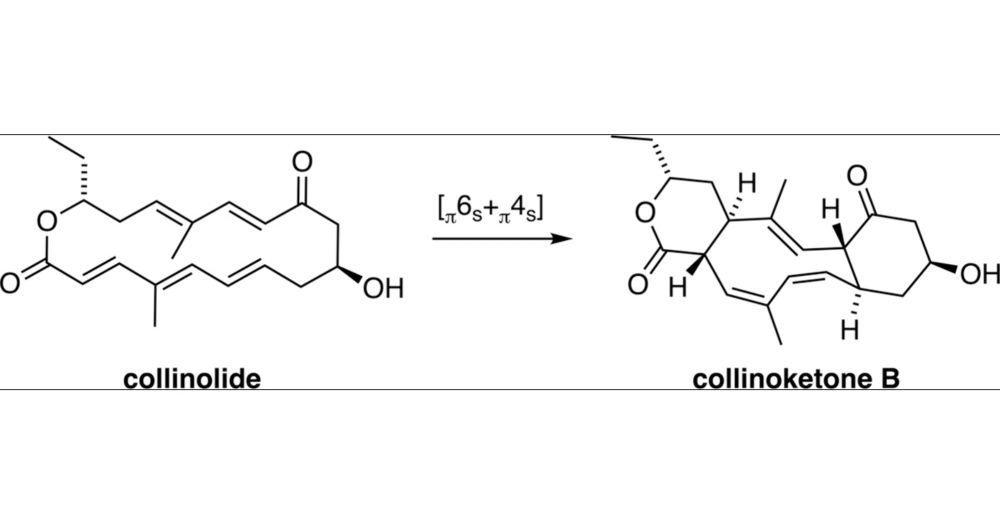
pubs.acs.org/doi/10.1021/...
a 🧵 1/n
Drain: arxiv.org/abs/2511.04820
Strain: direct.mit.edu/qss/article/...
Oligopoly: direct.mit.edu/qss/article/...




a 🧵 1/n
Drain: arxiv.org/abs/2511.04820
Strain: direct.mit.edu/qss/article/...
Oligopoly: direct.mit.edu/qss/article/...
🚀 Join ICIQ as a Junior Group Leader!
We’re seeking talented, innovative scientists ready to push the frontiers of research.
Apply now 👉🏼 careers.iciq.org/jobs/6745027...
@cerca.cat @bist.eu #SOMM_alliance

🚀 Join ICIQ as a Junior Group Leader!
We’re seeking talented, innovative scientists ready to push the frontiers of research.
Apply now 👉🏼 careers.iciq.org/jobs/6745027...
@cerca.cat @bist.eu #SOMM_alliance
Published annually by Pfizer Merck Takeda and others
Review of synthetic methods to marketed drugs
Great look at the current state of pharma synthesis

Published annually by Pfizer Merck Takeda and others
Review of synthetic methods to marketed drugs
Great look at the current state of pharma synthesis
#ChemSky
pubs.acs.org/doi/10.1021/...

#ChemSky
pubs.acs.org/doi/10.1021/...
Checkout our novel Ru-PIT system, just published in Chem!
Congratulations to all authors and a big thank you to Andrea Hamza for the wonderful collaboration!

Checkout our novel Ru-PIT system, just published in Chem!
Congratulations to all authors and a big thank you to Andrea Hamza for the wonderful collaboration!
www.nature.com/articles/s41...

www.nature.com/articles/s41...
Florian Doettinger, Jonathan Sagaya, Giacomo Morselli in @jacs.acspublications.org
pubs.acs.org/doi/10.1021/...

Florian Doettinger, Jonathan Sagaya, Giacomo Morselli in @jacs.acspublications.org
pubs.acs.org/doi/10.1021/...
Thank you, Angew. Chem. (open access): onlinelibrary.wiley.com/doi/10.1002/...

Thank you, Angew. Chem. (open access): onlinelibrary.wiley.com/doi/10.1002/...

pubs.acs.org/doi/10.1021/...

pubs.acs.org/doi/10.1021/...




