
@IRBBarcelona,bsky.social
This research was supported by the @erc.europa.eu, the Asociación Española Contra el Cáncer (AECC), @caixaresearch.bsky.social , Worldwide Cancer Research, La Marató de TV3, CIBERONC, AGAUR, Fundación Olga Torres, and the Spanish Ministry of Science and Innovation.

This research was supported by the @erc.europa.eu, the Asociación Española Contra el Cáncer (AECC), @caixaresearch.bsky.social , Worldwide Cancer Research, La Marató de TV3, CIBERONC, AGAUR, Fundación Olga Torres, and the Spanish Ministry of Science and Innovation.
We thank all members of the BatlleLab and our collaborators @irbbarcelona.org & @cnag-eu.bsky.social. Special mention to A. Riera and C. Sanchez for TGFβ inhibitor synthesis. We acknowledge the dedication of technical platforms, the bioinformatic unit, and the animal facility.

We thank all members of the BatlleLab and our collaborators @irbbarcelona.org & @cnag-eu.bsky.social. Special mention to A. Riera and C. Sanchez for TGFβ inhibitor synthesis. We acknowledge the dedication of technical platforms, the bioinformatic unit, and the animal facility.
Our work shows that TGFβ coordinates immune suppression across the adaptive and innate compartments, generating a two-layer defense that tumors use to resist immunotherapy.
Breaking this dual barrier — targeting TGFβ alongside PD-1/PD-L1 — can turn CRCs into immunotherapy-sensitive disease

Our work shows that TGFβ coordinates immune suppression across the adaptive and innate compartments, generating a two-layer defense that tumors use to resist immunotherapy.
Breaking this dual barrier — targeting TGFβ alongside PD-1/PD-L1 — can turn CRCs into immunotherapy-sensitive disease
TGFβ inhibition (via SMIs: galunisertib, LY3200882, or vactosertib) had dramatic effects on these models:
✅ It dismantled the collagen network produced by CAFs
✅It opened the path for peripheral CD8⁺ T cells to enter metastatic lesions
✅It made refractory tumors responsive to aPD-L1 therapy

TGFβ inhibition (via SMIs: galunisertib, LY3200882, or vactosertib) had dramatic effects on these models:
✅ It dismantled the collagen network produced by CAFs
✅It opened the path for peripheral CD8⁺ T cells to enter metastatic lesions
✅It made refractory tumors responsive to aPD-L1 therapy
We validated these findings in liver metastases generated by mouse tumor organoids carrying 3 or 4 driver mutations (Apc, Kras, Trp53, ±Tgfbr2 or Smad4).
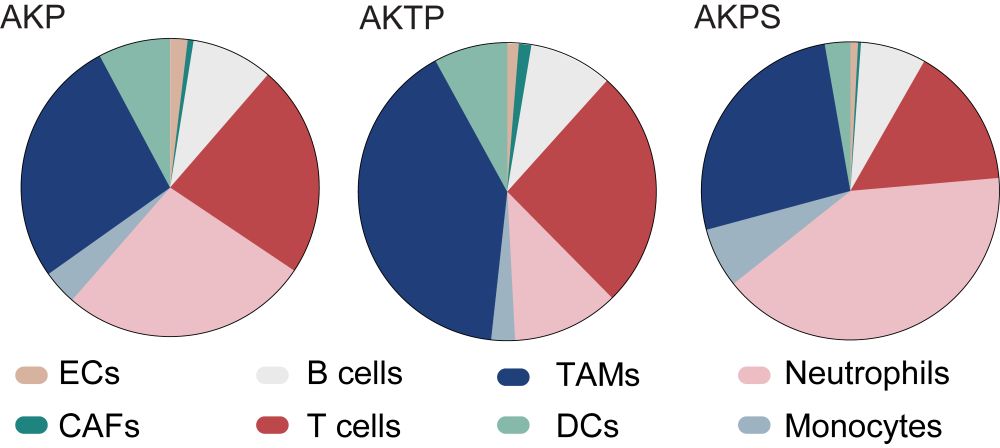
We validated these findings in liver metastases generated by mouse tumor organoids carrying 3 or 4 driver mutations (Apc, Kras, Trp53, ±Tgfbr2 or Smad4).
In the absence of Osteopontin - (Spp1 KO) - metastases remained T cell infiltrated and checkpoint blockade exerted potent therapeutic responses, suggesting therapeutic agents against Osteopontin, could be tested as a proxy for TGFβ inhibition.

In the absence of Osteopontin - (Spp1 KO) - metastases remained T cell infiltrated and checkpoint blockade exerted potent therapeutic responses, suggesting therapeutic agents against Osteopontin, could be tested as a proxy for TGFβ inhibition.
By generating macrophage-specific Tgfbr2 knockout mice, Ana Henriques showed that this population limits T cell proliferation in the TME.
📌Remarkably, we discovered that SPP1 production is necessary for this immunosuppressive macrophage response.

By generating macrophage-specific Tgfbr2 knockout mice, Ana Henriques showed that this population limits T cell proliferation in the TME.
📌Remarkably, we discovered that SPP1 production is necessary for this immunosuppressive macrophage response.
In parallel, single-cell profiling of the myeloid compartment revealed that TGFβ instructs an immunosuppressive macrophages population characterized by the expression of SPP1 (osteopontin).

In parallel, single-cell profiling of the myeloid compartment revealed that TGFβ instructs an immunosuppressive macrophages population characterized by the expression of SPP1 (osteopontin).
Meanwhile, aPD-L1 did something remarkable but incomplete: it boosted T cell motility allowing infiltrated cells to move and engage targets.
TGFβ inhibition enabled their entry but not their movement.
✅Only combination therapy achieved both, unleashing a fully active antitumor immune response
Meanwhile, aPD-L1 did something remarkable but incomplete: it boosted T cell motility allowing infiltrated cells to move and engage targets.
TGFβ inhibition enabled their entry but not their movement.
✅Only combination therapy achieved both, unleashing a fully active antitumor immune response
In experimental models of advanced colorectal cancer (CRC), we showed that TGFβ signaling on T cells prevented the influx of memory-like T cells from the lymph nodes into the metastatic TME.
🔴Without TGFβ inhibition, the number of T cells is insufficient to eradicate metastatic disease.

In experimental models of advanced colorectal cancer (CRC), we showed that TGFβ signaling on T cells prevented the influx of memory-like T cells from the lymph nodes into the metastatic TME.
🔴Without TGFβ inhibition, the number of T cells is insufficient to eradicate metastatic disease.
@hoheyn.bsky.social and @paulanietog.bsky.social identified diverse CD8⁺ T cell states — from naïve-like and progenitor to effector and exhausted — and together with Maria Salvany-Celades, they traced how TGFβ inhibition restores clonal expansion and effector differentiation.

@hoheyn.bsky.social and @paulanietog.bsky.social identified diverse CD8⁺ T cell states — from naïve-like and progenitor to effector and exhausted — and together with Maria Salvany-Celades, they traced how TGFβ inhibition restores clonal expansion and effector differentiation.
To understand the immune landscape at single-cell resolution, @hoheyn.bsky.social and @paulanietog.bsky.social from @cnag-eu.bsky.social led an extensive scRNA-seq + TCR-seq analysis of tumor-infiltrating lymphocytes (TILs) and myeloid cells in models of metastatic CRC under treatment.
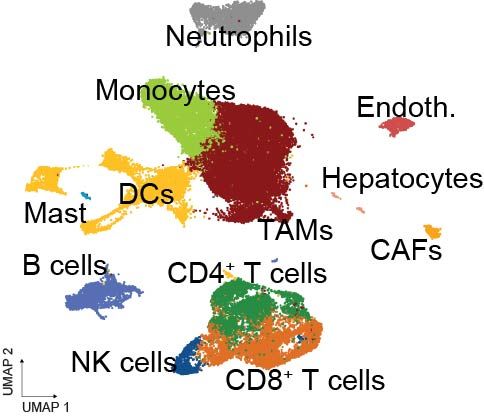
To understand the immune landscape at single-cell resolution, @hoheyn.bsky.social and @paulanietog.bsky.social from @cnag-eu.bsky.social led an extensive scRNA-seq + TCR-seq analysis of tumor-infiltrating lymphocytes (TILs) and myeloid cells in models of metastatic CRC under treatment.
👌A tremendous team effort led by Ana Henriques, Maria Salvany-Celades, & Alejandro Prados in the BatlleLab, in collaboration with @paulanietog.bsky.social and @hoheyn.bsky.social, with the help of amazing multidisciplinary teams at @irbbarcelona.org, and @cnag-eu.bsky.social.
Kudos to all 🔝🔝🙌🙌

👌A tremendous team effort led by Ana Henriques, Maria Salvany-Celades, & Alejandro Prados in the BatlleLab, in collaboration with @paulanietog.bsky.social and @hoheyn.bsky.social, with the help of amazing multidisciplinary teams at @irbbarcelona.org, and @cnag-eu.bsky.social.
Kudos to all 🔝🔝🙌🙌
🔝 A huge thank you to the organizers for the invitation, and for putting together such a memorable conference.
📸 A remarkable aspect was seeing together five generations of Clevers’ Lab!!

🔝 A huge thank you to the organizers for the invitation, and for putting together such a memorable conference.
📸 A remarkable aspect was seeing together five generations of Clevers’ Lab!!
You’ll gain new insights into fundamental mechanisms controlling cancer cell plasticity as well as innovative approaches to target them
➡️https://eacr.org/conference/cancercellplasticity2025
@helloeacr.bsky.social

You’ll gain new insights into fundamental mechanisms controlling cancer cell plasticity as well as innovative approaches to target them
➡️https://eacr.org/conference/cancercellplasticity2025
@helloeacr.bsky.social
CRC organoids under treatment with KRAS inhibitor RMC-9945, rapidly acquire a LGR5+ cancer stem cell-like phenotype, allowing them to bypass KRAS inhibition, and eventually resume growth.
Centonze et al., in #CancerDiscovery
#AACR
CRC organoids under treatment with KRAS inhibitor RMC-9945, rapidly acquire a LGR5+ cancer stem cell-like phenotype, allowing them to bypass KRAS inhibition, and eventually resume growth.
Centonze et al., in #CancerDiscovery
#AACR
Last but not least, we acknowledge our funding sources.
This research was supported by the European Research Council (ERC), Spanish Association Against Cancer (AECC), La Caixa Foundation, CIBERONC, Horizon Europe, Revolution Medicines and the Spanish Ministry of Science and Innovation.

Last but not least, we acknowledge our funding sources.
This research was supported by the European Research Council (ERC), Spanish Association Against Cancer (AECC), La Caixa Foundation, CIBERONC, Horizon Europe, Revolution Medicines and the Spanish Ministry of Science and Innovation.
We are deeply grateful to Revolution Medicines, both for great input, research support and for the invaluable opportunity to work with the KRAS-G12D inhibitor RMC-9945.


We are deeply grateful to Revolution Medicines, both for great input, research support and for the invaluable opportunity to work with the KRAS-G12D inhibitor RMC-9945.
📌 Key takeaway message:
KRAS is not only an oncogenic driver — it is also a cell-state regulator.
By mapping this plasticity, we identify new therapeutic angles and combination strategies for KRAS-mutant metastatic CRC.

📌 Key takeaway message:
KRAS is not only an oncogenic driver — it is also a cell-state regulator.
By mapping this plasticity, we identify new therapeutic angles and combination strategies for KRAS-mutant metastatic CRC.
Our results reveal a functional interplay: upon KRAS inhibition, CRC cells lean on WNT/β-catenin signaling for survival and growth.
➡️ We thus tested a combination approach: KRAS-G12D inhibition plus targeted removal of LGR5⁺ cells, which led to stronger tumor control and prolonged survival.

Our results reveal a functional interplay: upon KRAS inhibition, CRC cells lean on WNT/β-catenin signaling for survival and growth.
➡️ We thus tested a combination approach: KRAS-G12D inhibition plus targeted removal of LGR5⁺ cells, which led to stronger tumor control and prolonged survival.
Mechanistically, Marc Marti-Renom and Meritxell Novillo-Font (CNAG-CRG) analysis of 3D chromatin and TF occupancy studies showed a switch in transcriptional control, without the need for major chromatin reorganization:
• AP-1 factors (HRC-linked) ↓
• TCF/LEF factors (WNT/CSC-linked) ↑
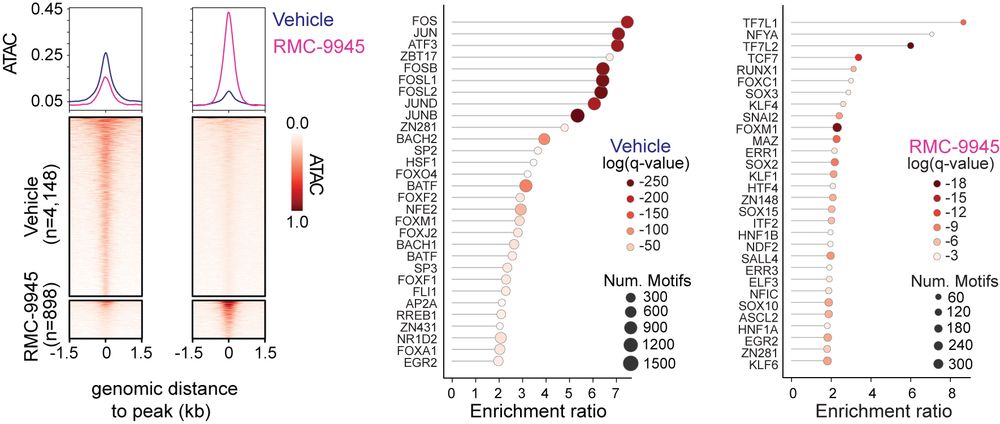
Mechanistically, Marc Marti-Renom and Meritxell Novillo-Font (CNAG-CRG) analysis of 3D chromatin and TF occupancy studies showed a switch in transcriptional control, without the need for major chromatin reorganization:
• AP-1 factors (HRC-linked) ↓
• TCF/LEF factors (WNT/CSC-linked) ↑
Hector Palmer and Jordi Martinez-Quintanilla (VHIO) made equivalent observations in BRAF mutant CRCs treated with encorafenib plus cetuximab, implying overactivation of the MAPK pathway promotes plasticity towards the EMP1+ HRC state.

Hector Palmer and Jordi Martinez-Quintanilla (VHIO) made equivalent observations in BRAF mutant CRCs treated with encorafenib plus cetuximab, implying overactivation of the MAPK pathway promotes plasticity towards the EMP1+ HRC state.
Upon KRAS inhibition with RMC-9945, we observed a striking and rapid cell-state reprogramming in pre-clinical liver metastasis models and Patient-derived organoids: metastases naturally enriched in EMP1⁺ High-Relapse Cells (HRCs) transition to an LGR5⁺ canonical cancer stem-like state.

Upon KRAS inhibition with RMC-9945, we observed a striking and rapid cell-state reprogramming in pre-clinical liver metastasis models and Patient-derived organoids: metastases naturally enriched in EMP1⁺ High-Relapse Cells (HRCs) transition to an LGR5⁺ canonical cancer stem-like state.
KRAS mutations are present in ~40% of colorectal cancers, and KRAS-targeted inhibitors are now entering the clinic.
❓This led us to a key question:
➡️ What happens to HRC-rich metastases when we pharmacologically block KRAS activity?

KRAS mutations are present in ~40% of colorectal cancers, and KRAS-targeted inhibitors are now entering the clinic.
❓This led us to a key question:
➡️ What happens to HRC-rich metastases when we pharmacologically block KRAS activity?
HRCs or High-Relapse Cells, marked by EMP1 expression, are found at invasive fronts and are associated with poor prognosis. Other labs have identified non-LGR5+ states (named oncofetal or regenerative), also marked by EMP1. We showed that EMP1+ cells are linked to KRAS activating mutations.

HRCs or High-Relapse Cells, marked by EMP1 expression, are found at invasive fronts and are associated with poor prognosis. Other labs have identified non-LGR5+ states (named oncofetal or regenerative), also marked by EMP1. We showed that EMP1+ cells are linked to KRAS activating mutations.

