
www.computerworld.com/article/4059...

www.computerworld.com/article/4059...


Et quand on les évalue correctement...

Et quand on les évalue correctement...
INSPECT-SR: A tool for assessing trustworthiness of randomised controlled trials.

INSPECT-SR: A tool for assessing trustworthiness of randomised controlled trials.
Par @olivierhertel.bsky.social

Par @olivierhertel.bsky.social

I'm just saying that generating a hypothesis from the entirety of human knowledge that preceded your data is a much safer bet.
I'm just saying that generating a hypothesis from the entirety of human knowledge that preceded your data is a much safer bet.
Retracted papers are included in meta-analyses published in HIF journal. Inclusion of the retracted studies changed
effect estimates.
jamanetwork.com/journals/jam...
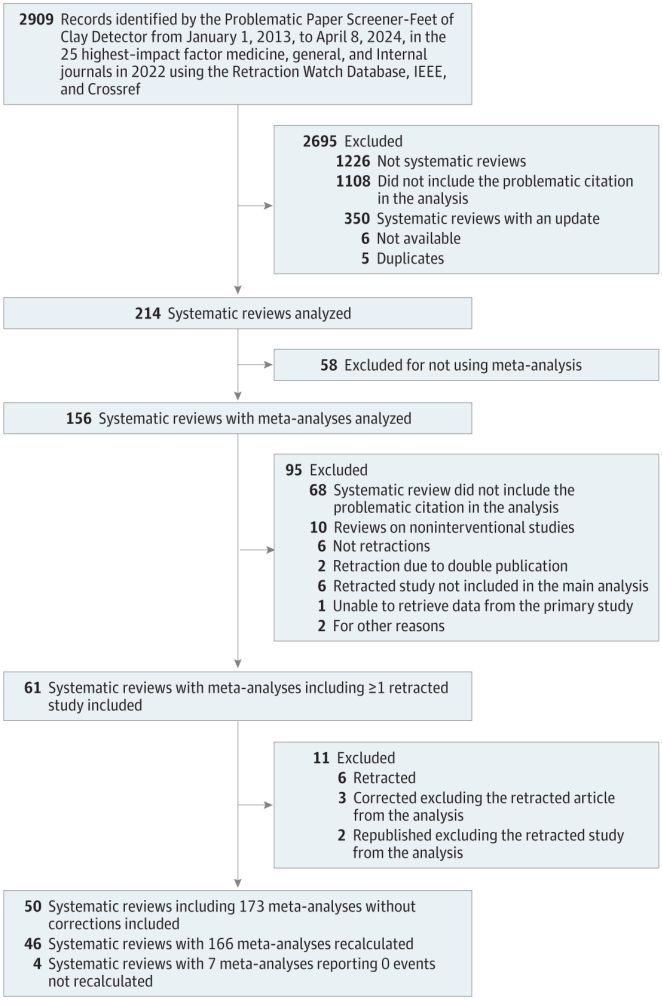
Retracted papers are included in meta-analyses published in HIF journal. Inclusion of the retracted studies changed
effect estimates.
jamanetwork.com/journals/jam...
link.springer.com/article/10.1...

link.springer.com/article/10.1...



Still no proper response from the journal (other then many "we'll look into it"). It's been a year now.
Our paper "A Comparison of Statistical Methods for Time-To-Event Analyses in Randomized Controlled Trials Under Non-Proportional Hazards" got published today 🎉
We describe commonly used methods, and compare their performance in a simulation study across different scenarios.

Our paper "A Comparison of Statistical Methods for Time-To-Event Analyses in Randomized Controlled Trials Under Non-Proportional Hazards" got published today 🎉
We describe commonly used methods, and compare their performance in a simulation study across different scenarios.
ja.ma/3ENOh4U
#MedSky

ja.ma/3ENOh4U
#MedSky
María Villalobos-Quesada, a researcher at Leiden University Medical Centre, discusses the need for transparent and consistent reporting of quality for #AI tools with JAMA+ AI.
#MedSky


"Uncertainty of risk estimates from clinical prediction models: rationale, challenges, and approaches"
- most models provide just a risk estimate
- we argue for presenting associated uncertainty too
- includes pros, cons, PPIE & methods
Hope helpful!
www.bmj.com/content/388/...

"Uncertainty of risk estimates from clinical prediction models: rationale, challenges, and approaches"
- most models provide just a risk estimate
- we argue for presenting associated uncertainty too
- includes pros, cons, PPIE & methods
Hope helpful!
www.bmj.com/content/388/...

