@erasmusmc.bsky.social 🎉📢 whose complementary work led by Lanpeng Chen and Yujie Bian revealed similar inflammatory bone marrow remodeling in MDS and made a link to leukemic evolution. A story that unfolded in parallel! ✏️
shorturl.at/4G8Yr

@erasmusmc.bsky.social 🎉📢 whose complementary work led by Lanpeng Chen and Yujie Bian revealed similar inflammatory bone marrow remodeling in MDS and made a link to leukemic evolution. A story that unfolded in parallel! ✏️
shorturl.at/4G8Yr


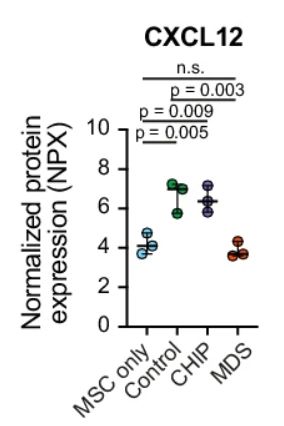




The scale of human stromal niche profiling is what makes this study special! 🦴🩸🧬🔬


The scale of human stromal niche profiling is what makes this study special! 🦴🩸🧬🔬
@unimainz.bsky.social @tudresden.bsky.social @dkfz.bsky.social @trowbridgelab.bsky.social @ki.se
@unimainz.bsky.social @tudresden.bsky.social @dkfz.bsky.social @trowbridgelab.bsky.social @ki.se







The scale of human stromal niche profiling is what makes this study special! 🦴🩸🧬🔬


The scale of human stromal niche profiling is what makes this study special! 🦴🩸🧬🔬
I hope you like it!
www.biorxiv.org/content/10.1...

I hope you like it!
www.biorxiv.org/content/10.1...

